Recently, researchers from the Catalysis and New Materials Research Center (Group 1500) of DICP, including Academician Tao Zhang, Prof. Xiaodong Wang, Assoc. Prof. Chuande Huang, and Assoc. Prof. Botao Qiao, have developed an enzyme-mimicking copper-based single-atom catalyst (Cu1/CN) that enables the efficient conversion of methane into oxygenated compounds.
The direct selective oxidation of methane into value-added oxygenates (e.g., methanol, methyl hydroperoxide) is considered a crucial pathway for the rational utilization of natural gas resources. However, because oxygenates are typically more reactive than methane and possess higher molecular polarity, they tend to be readily adsorbed and activated on catalyst surfaces, leading to over-oxidation and thus lowering the selectivity toward target products. Therefore, the design of highly efficient catalysts that can simultaneously achieve high methane conversion and high product selectivity remains a major research challenge.
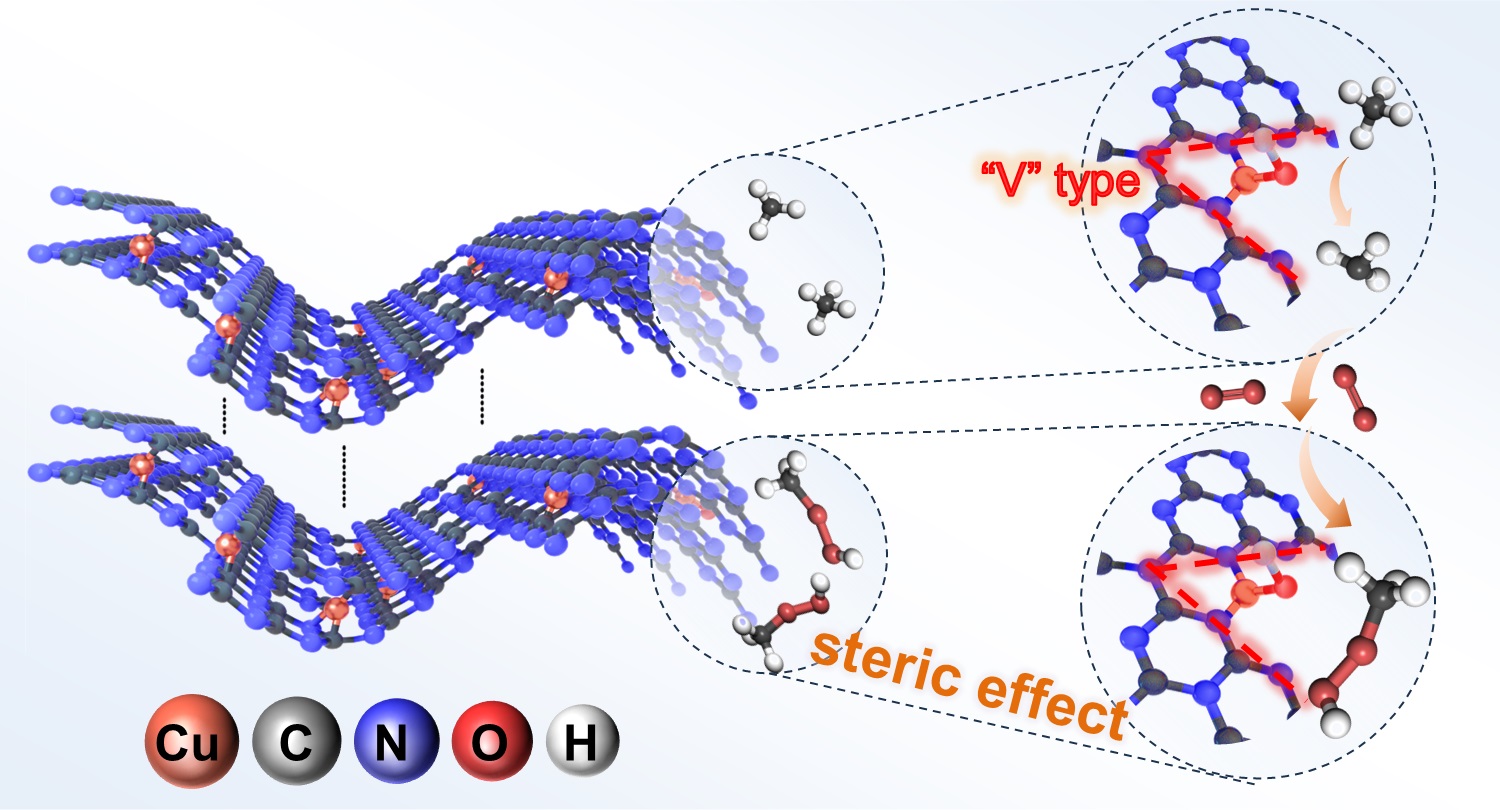
In this work, inspired by the active structure and working mechanism of natural methane monooxygenases (MMOs), the researchers anchored single-atom copper (Cu1) on the “zigzag” edges of graphitic carbon nitride (C3N4) to construct a “V”-shaped N2–Cu1–O active center. It was found that the Cu1–O center could efficiently activate methane via a hydrogen abstraction pathway with an energy barrier of only 0.58 eV, generating methyl radicals (•CH3). Under reaction conditions at 50 °C, the process exhibited an outstanding catalytic activity, with a turnover frequency (TOF) of 405.3 ± 8.2 h−1. The resulting •CH3 radicals were subsequently captured by molecular oxygen (O2) in the reaction system, leading predominantly to the formation of methyl hydroperoxide (CH3OOH) as the oxygenated product. Moreover, the unique “V”-shaped configuration of the catalytic site provides a molecular channel of 3.87 Å, which readily accommodates methane molecules (kinetic diameter: 3.78 Å) while effectively preventing the re-adsorption of CH3OOH (kinetic diameter: 4.23 Å) onto the Cu1 center. This structural feature suppresses over-oxidation and ensures a product selectivity of nearly 100%.
In recent years, Prof. Xiaodong Wang’s team has been devoted to the selective oxidation of light alkanes (Energy Environ. Sci., 2019;Appl. Catal. B-Environ., 2020; ACS Catal., 2020; Nat. Commun., 2021; Nat. Catal., 2022; Nat. Commun., 2024). Building upon their earlier advances in single-atom catalysis for the selective oxidation of methane (Angew. Chem. Int. Ed., 2022; J. Am. Chem. Soc., 2023; Angew. Chem. Int. Ed., 2024; Appl. Catal. B-Environ., 2025), this work further exploits the spatial confinement effect of the support to achieve the selective oxidation of methane to high-value-added chemicals under mild and low-cost conditions. This study offers new insights into the design of highly efficient single-atom catalysts.
The related findings, entitled “Enzyme-mimicking copper single-atom catalyst for selective oxidation of methane to liquid oxygenates”, were recently published in the Journal of the American Chemical Society . This work was supported by the NSFC Basic Science Center Program for “Single-Atom Catalysis”, the National Natural Science Foundation of China, the Youth Innovation Promotion Association of CAS, and other funding programs. (Text/Images: Chuande Huang)
Publication Link: https://pubs.acs.org/doi/10.1021/jacs.5c10016